PDF) Efficacy and safety of DFN-11 (sumatriptan injection, 3 mg) in adults with episodic migraine: An 8-week open-label extension study

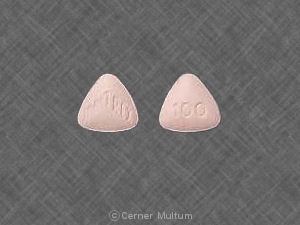
These highlights do not include all the information needed to use SUMATRIPTAN INJECTION safely and effectively. See full prescribing information for SUMATRIPTAN INJECTION. SUMATRIPTAN injection, for subcutaneous useInitial U.S. Approval: 1992

PDF) DFN-02, Sumatriptan 10 mg Nasal Spray with Permeation Enhancer, for the Acute Treatment of Migraine: A Randomized, Double-Blind, Placebo-Controlled Study Assessing Functional Disability and Subject Satisfaction with Treatment

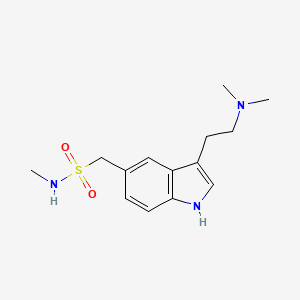
US20060002989A1 - Formulations of sumatriptan for absorption across biological membranes, and methods of making and using the same - Google Patents

Dabigatran Market to Witness Huge Growth by 2028 | Pfizer, Bayer Healthcare, GlaxoSmithKline, Boehringer Ingelheim – Energy Siren